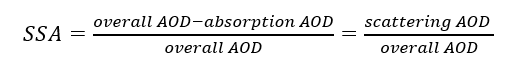1. Size
Aerosols in the air scatter and absorb the sunlight.
Aerosols vary in size from 0.001μm to 100μm, and most of them are sorted as accumulation mode(0.1~1μm). These small particles have long residence time; they remain in the air for weeks, mainly scattering sunlight. However, larger aerosols such as black carbon(BC) and dust particles have shorter residence time from hours to days, thus having shorter time to attenuate solar radiation. Larger particles readily absorb sunlight rather than reflect it, which has warming effect on the atmosphere.
In conclusion, the higher the proportion of small particles in entire aerosols in the air (i.e., the higher the FF(Fine Fraction)), the more sunlight the atmosphere scatters, resulting in lower atmospheric temperature.
Aerosols vary in size from 0.001μm to 100μm, and most of them are sorted as accumulation mode(0.1~1μm). These small particles have long residence time; they remain in the air for weeks, mainly scattering sunlight. However, larger aerosols such as black carbon(BC) and dust particles have shorter residence time from hours to days, thus having shorter time to attenuate solar radiation. Larger particles readily absorb sunlight rather than reflect it, which has warming effect on the atmosphere.
In conclusion, the higher the proportion of small particles in entire aerosols in the air (i.e., the higher the FF(Fine Fraction)), the more sunlight the atmosphere scatters, resulting in lower atmospheric temperature.
|
*
Aerosol with high hygroscopic property is easily attached to moisture and inflates its size (e.g. condensation nucleus). Larger aerosols result in better absorption of solar radiation, thus the overall AOD increases as the absorption AOD increases. However, since we care more about scattering AOD rather than overall AOD in this study, we don't need to consider that larger aerosols are more effective in lowering the atmospheric temperature only because they result in high overall AOD. |
2. Optical Properties
The optical properties of aerosols regarding their chemical components are another factor that have impact on AOD. How much light an aerosol scatters and absorbs can be seen respectively by its scattering and absorption coefficient. When comparing soluble ions (e.g. sulfate, nitrate), BC, and sea-salt particles, soluble ions generally have the highest scattering coefficient value, and BC has the highest absorption coefficient value. This is because of the property of BC that absorbs visible and near-infrared rays of solar radiation. And the absorption coefficient of sea-salt particles is very small compared to that of other components.
In 2013 and 2014, the scattering coefficients of the water-soluble component in Gwangjin-gu, Seoul were 140 Mm^-1 and 154 Mm^-1, respectively, indicating higher values than other components. In addition, although BC concentrations at Aewol, Jeju in 2013 and 2014 were the lowest among the particles, BC had the highest absorption coefficients of 5.6 Mm^-1 and 6.7 Mm^-1, respectively. Meanwhile, the absorption coefficient by the sea-salt at all points was very small (<0.001 Mm^-1).
Yun-Yeong So, Sang-Keun Song, Yu-Na Choi, Comparison of the concentration characteristics and optical properties of aerosol chemical components in different regions, Journal of environmental Science International Vol.28 No.1 pp.107-123
In the process of finding aerosols that efficiently scatter sunlight, we will introduce the term, Single Scattering Albedo (SSA), which is defined as (scattering AOD)/(overall AOD). In other words, the more scattering aerosols exist in the air compared to absorbing aerosols, the closer the SSA gets to its maximum value, which is 1. As SSA increases, the atmosphere scatters more solar radiation, thus the atmospheric temperature decreases.
Since the purpose of the climate engineering is to lower the temperature of the atmosphere, we should use aerosols that make SSA as close to 1 as possible to increase the utility of the climate engineering. We found earlier that substances with high FF scatter more sunlight than those with low FF, and soluble ions scatter more sunlight than other substances. Therefore, a substance that satisfies both conditions would be an ideal aerosol that we are looking for.